STERILE INJECTABLES RPO AND RECRUITING SERVICES
Specialized recruiting for sterile injectables and CDMO talent needs
From aseptic fill-finish to clean room production, we help CDMOs and pharma manufacturers build high-performing teams for high-compliance environments.

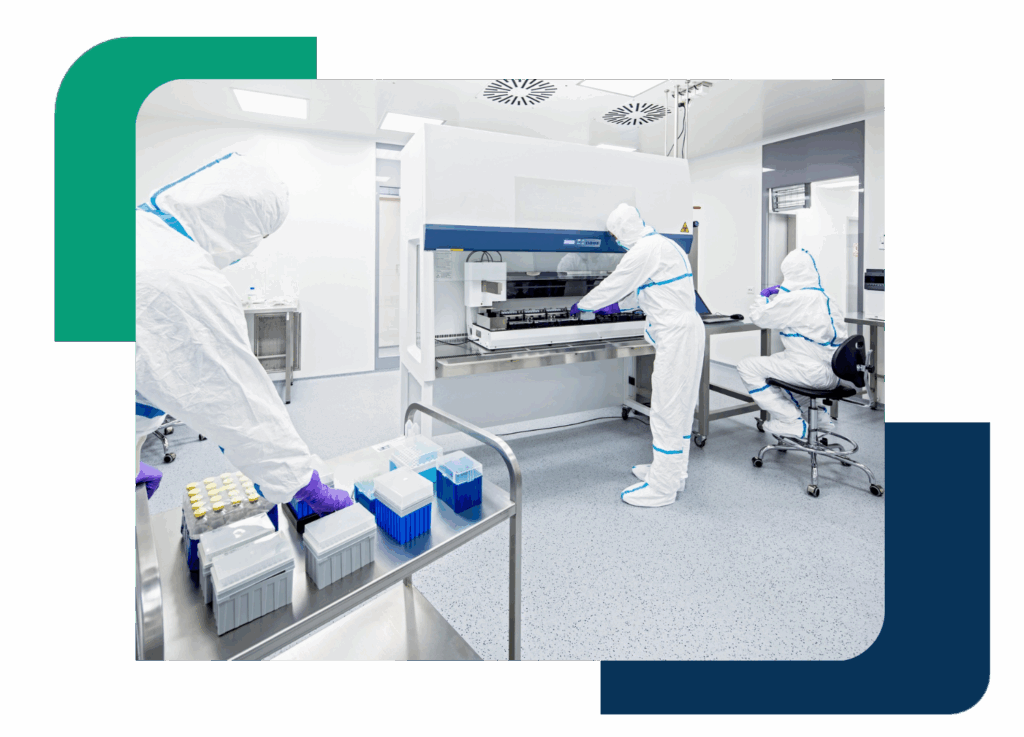
Recruiting for high-stakes sterile environments
Hiring for sterile manufacturing comes with unique challenges. We’re built to solve them.
Here are just a few of the pain points we help our clients solve:
- Sourcing talent for aseptic fill-finish operations
- Staffing clean room environments with high turnover risk
- Finding candidates who can follow strict gowning and decontamination procedures
- Addressing time-to-fill delays due to long onboarding or training cycles
- Scaling rapidly for new client partnerships or tech transfer projects
- Meeting cGMP compliance without compromising on quality or culture fit
We understand what’s at stake and how to find the talent that keeps your operations running smoothly.
Purpose-built recruiting for complex manufacturing
We don’t rely on off-the-shelf candidate pools. Instead, we custom-build pipelines aligned to the unique demands of your operation—whether you’re expanding capacity, launching a tech transfer, or scaling fill-finish capabilities.
- Sourcing aligned to your environment – We start by understanding your facility requirements, workflow constraints, and role-specific challenges.
- Deeper candidate evaluation – Beyond qualifications, we screen for mindset, process discipline, and long-term fit in GMP-regulated settings.
- Experience with regulated manufacturing environments – Our recruiters know how to navigate strict operational and documentation requirements across pharma and life sciences.
Real-world success: Hiring for clean room conditions

The challenge:
A client needed to hire for a clean room environment where employees had to fully gown up, pass through air filtration, and follow strict protocols, just to start their shift. Even taking a bathroom break meant repeating a 40+ minute procedure.
Our solution:
We customized our sourcing and screening approach to identify candidates who could handle both the physical and psychological demands. Some had relevant experience; others demonstrated the mindset and discipline to adapt and succeed.
The result:
The client saw reduced turnover, better retention, and improved productivity—all without compromising quality or compliance.
Move faster without sacrificing quality
Sterile manufacturing roles—including those in clean rooms, aseptic production, and sterile packaging—take, on average, 13 days longer to fill than general pharma positions.
In CDMO environments where timelines are tight and capacity matters, delays can disrupt client delivery and operational throughput.
With Advanced RPO, you gain a recruiting partner that delivers speed, precision, and transparency at every step.


Let’s connect and explore how we can support your sterile manufacturing workforce needs.
Not ready to connect? Start with these resources.
Successful recruiting in sterile injectables and CDMO environments starts with knowing which metrics move the needle. See how industry leaders are measuring performance—and where your benchmarks should be.

